The modern era of polymer science belongs to the chemical engineer. Over the years, polymer chemists have invented a wealth of novel macromolecules and polymers. Yet understanding how these molecules can be synthesized and processed to exhibit their maximum theoretical properties is still a frontier for research. Only recently has modern instrumentation been developed to help us understand the fundamental interactions of macromolecules with themselves, with particulate solids, with organic and inorganic fibers, and with other surfaces. Chemical engineers are using these tools to probe the microscale dynamics of macromolecules. Using the insight gained from these techniques, they are manipulating macromolecular interactions both to develop improved processes and to create new materials.
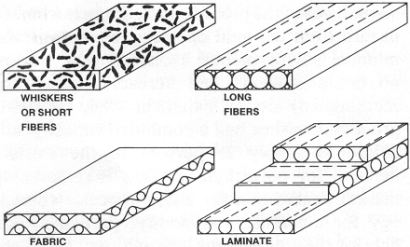
The power of chemical processing for controlling materials structure on the microscale is illustrated by the current generation of high-strength polymer fibers, some of which have strength-to-weight ratios an order of magnitude greater than steel ("Order of magnitude" is synonymous with "a factor of 10." Each additional order of magnitude corresponds to an additional factor of 10, so, for example, 3 orders of magnitude is 10x10x10 = 1000 times). One of the best known example of these fibers, Kevlar, is prepared by spinning an aramid polymer—part of the family of nylons—from an anisotropic phase (a liquid phase in which molecules are spontaneously oriented with one another over microscopic dimensions). This spontaneous orientation is the result of both the processing conditions chosen and the highly rigid linear molecular structure of the aramid polymer. During spinning, the oriented regions in the liquid phase align with the fiber axis to give the resulting fiber high strength and rigidity. The concept of spinning fibers from anisotropic phases has been extended to both solutions and melts of newer polymers, such as polybenzothiazole, as well as traditional polymers such as polyethylene. Ultrahigh-strength fibers of polyethylene have been prepared by gel spinning. The same concept, controlling the molecular orientation of polymers to produce high strength, is also being achieved through other processes, such as fiber-stretching carried out under precise conditions.
|
What is a
polymer?
The word comes from the greek
polumeres, which means "having many parts."
Polymers are large molecules consisting of repeated
chemical units ('mers') joined together, usually in
a line, like beads on a string. Each 'mer' is
typically made up of more than 5 and less than 500
atoms; the word "polymer" is applied when you have
more than about 50 'mers' stuck together. Most of
the plastics that make up the pieces of junk that
fill our lives are made of polymers.
Historically, polymers have
mostly been used to make solid plastics where the
chains virtually don't move. But nowadays people
dream of new applications of polymer liquids where
microscopic random fluctuations (Brownian motion)
and interactions (the sticking together or
association of different types of molecules) can
play a more important role. Many of the most
important research problems involve polymers free
to fluctuate about in a small-molecule solvent.
Naturally, the most important solvent is also the
hardest one to understand: water. An important area
of research is the modification of the properties
of surfaces using thin polymer coatings.
The sky is the limit for
these wet technologies: living organisms are mainly
composed of polymerized amino acids (proteins)
nucleic acids (RNA and DNA), and other biopolymers.
Synthetic polymers are
produced by chemical reactions, termed
"polymerizations." Polymerizations occur in a
variety of ways, but the common thread is reactions
consisting of the repetitive chemical bonding of
individual molecules, or monomers. Assorted
combinations of heat, pressure, and catalysis alter
the chemical bonds that hold monomers together,
causing them to bond with one another. Most often,
they do so in a linear fashion, creating chains of
monomers called polymers. Some polymerizations join
entire monomers together, whereas others join only
portions of monomers and create "leftover"
materials, or by-products. Co-polymers can be
formed using two or more different monomers. And
two or more polymers can be combined to produce an
alloy, or blend, that displays characteristics of
each component.
|
In addition to processes that result in materials with specific high-performance properties, chemical engineers continue to design new processes for the low-cost manufacture of polymers. The UNIPOL process for the manufacture of polyethylene is a good example of the contributions of engineering research to polymer processing. Polyethylene is probably the quintessential commodity polymer. It has been manufactured worldwide for decades, and current U.S. production exceeds 20 billion pounds per year.
|
Linear polyethylene consists of an large number
of CH2 units linked together.
|
Considering the global capital investment in existing plants for making polyethylene, it could be argued that inventing a new process for its manufacture is a waste of time and money. Not so. Chemical engineers at Union Carbide designed a proprietary catalyst that allowed polyethylene to be made in a fluidized-bed, gas-phase reactor operating at low temperature and pressure (below 100 ºC and 21 atmospheres pressure). The resulting process produces a polymer with exceptional uniformity and can precisely control the molecular weight and density of the product. The advantages of the process (including a low safety hazard from the mild operating conditions and minimal environmental impact since there are no liquid effluents and unreacted gases are recirculated) are such that, in 1986, UNIPOL process licensees had a combined capacity sufficient to supply 25 percent of the world's demand for polyethylene. This is remarkable market penetration for a new process technology for a mature commodity, particularly in light of the tremendous existing (and fully amortized) worldwide capacity for polyethylene. In 1985, Union Carbide and Shell Chemical successfully extended the UNIPOL process to the manufacture of polypropylene, another major polymer commodity. Interestingly, the first two licensees for the new polypropylene process were a Japanese chemical company and a Korean petroleum company.